When water boils do the bubbles tend to rise or sink?
As the heated fluid reaches its boiling point, the bubbles do not rise to the surface. Instead, the bubbles that do form coalesce into one large bubble that sits on the heated surface.
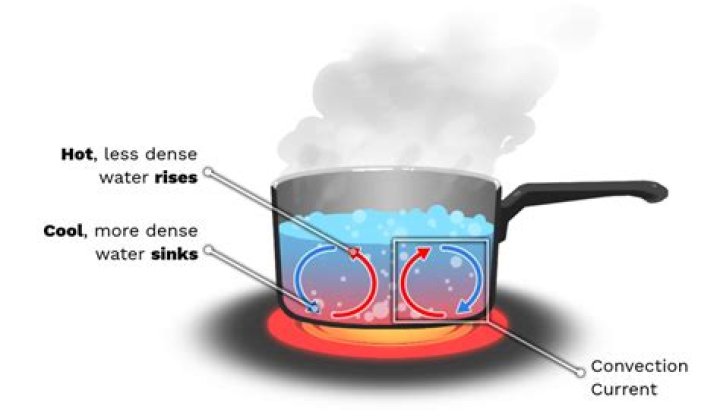
Which phase of water is densest?
Liquid water
[Liquid water is densest, water vapor is least dense.]
What happens when the water boils?
When boiling occurs, the more energetic molecules change to a gas, spread out, and form bubbles. These rise to the surface and enter the atmosphere. It requires energy to change from a liquid to a gas (see enthalpy of vaporization). In addition, gas molecules leaving the liquid remove thermal energy from the liquid.
What happens to water molecules when it boils?
When water is boiled, the heat energy is transferred to the molecules of water, which begin to move more quickly. Eventually, the molecules have too much energy to stay connected as a liquid. When this occurs, they form gaseous molecules of water vapor, which float to the surface as bubbles and travel into the air.
Does boiling remove oxygen from water?
During the boiling process bubbles of water vapour, depleted in oxygen, are produced and it is with these that gas exchange takes place. Dissolved oxygen is entrained in the bubbles and then liberated to the atmosphere at the liquid surface.
What is inside of bubbles when water boils?
These bubbles are water vapor. When you see water at a “rolling boil,” the bubbles are entirely water vapor. Water vapor bubbles start to form on nucleation sites, which are often tiny air bubbles, so as water starts to boil, the bubbles consist of a mixture of air and water vapor.
What phase of water is least densest?
Despite the “hardness” of ice, the spacing of water molecules per unit volume is actually greater than it is for liquid water. Hence, ice is less dense than liquid water (which is why ice cubes float).
What are two characteristics of Earth allow liquid water to exist?
The two characteristics are suitable atmospheric pressure and temperature. If the pressure is too low the water will boil of straight away.
What comes out when the water boils?
When water is heated it evaporates, which means it turns into water vapor and expands. At 100℃ it boils, thus rapidly evaporating. And at boiling point, the invisible gas of steam is created. The opposite of evaporation is condensation, which is when water vapor condenses back into tiny droplets of water.
Does salt help water boil?
One particularly stubborn myth is that adding salt will make the water take longer to come to a boil. Chemically speaking, it’s true that salt raises the boiling point; however, the amount of salt used in cooking applications is so small that it won’t make a difference with timing.
Why does a bubble form at the top of boiling water?
At the top surface, a bubble can either break and release the vapor into the air, or, if the temperature is low enough, the bubble can shrink. The temperature at the surface of boiling water may be cooler than the lower liquid because of the energy that is absorbed by water molecules when they change phases.
Is it possible to boil water without air bubbles?
As the temperature gets closer to the boiling point of the liquid, the bubbles will be the vapor phase of the substance. While you can boil water without air bubbles simply by reboiling it, you can’t reach the boiling point without getting vapor bubbles.
Why are steam bubbles less dense than water?
Because steam is less dense than water, steam bubbles are buoyed to the top of the water. If you’re merely heating the water without boiling it, the bubbles could be filled with dissolved gases that are escaping. The explanation is still the same, though; the gas bubbles are less dense than the surrounding liquid, so they rise.
Why do bubbles form in a swimming pool?
You can see this effect more clearly if you blow bubbles underwater in a swimming pool. The bubbles are much larger by the time they reach the surface. The water vapor bubbles start out larger as the temperature gets higher because more liquid is being converted to gas.



