What is the density of uranium?
about 19 grams per cubic centimeter
Density – Uranium is very dense. At about 19 grams per cubic centimeter, it is 1.67 times more dense than lead.
How much does Uranium 235 weigh?
Uranium-235
| General | |
|---|---|
| Isotope mass | 235.0439299 u |
| Spin | 7/2− |
| Excess energy | 40914.062 ± 1.970 keV |
| Binding energy | 1783870.285 ± 1.996 keV |
Why is there more uranium 238 than 235?
The U-238 nucleus also has 92 protons but has 146 neutrons – three more than U-235 – and therefore has a mass of 238 units. The difference in mass between U-235 and U-238 allows the isotopes to be separated and makes it possible to increase or “enrich” the percentage of U-235.
What is the mass of Uranium 235 in grams?
Properties of Uranium-235 Molecular Weight: The molecular weight of this metal is 235.044 g/mol. Atomic Number: The atomic number for this metal is 92. Atomic Mass: Its mass number is 235.0439299 u (unified atomic mass units).
How many atoms are in 1 kg of uranium?
so the answer is 2.56*10^24 atoms.
Can you eat 1 gram of uranium?
What happens if you eat a gram of uranium? The metal would react with the acid in your stomach, making you burp hydrogen. Consuming more, however, can either kill you or leave you susceptible to intestinal and stomach cancer.
What is the concentration of 235 U in uranium?
Natural Uranium – contains a 238 U concentration of 99.27 percent, 235 U concentration of 0.711 percent and very little 234 U. Low Enriched Uranium – contains a 235 U concentration between 0.711 percent and 20 percent.
What is the density of uranium in cm3?
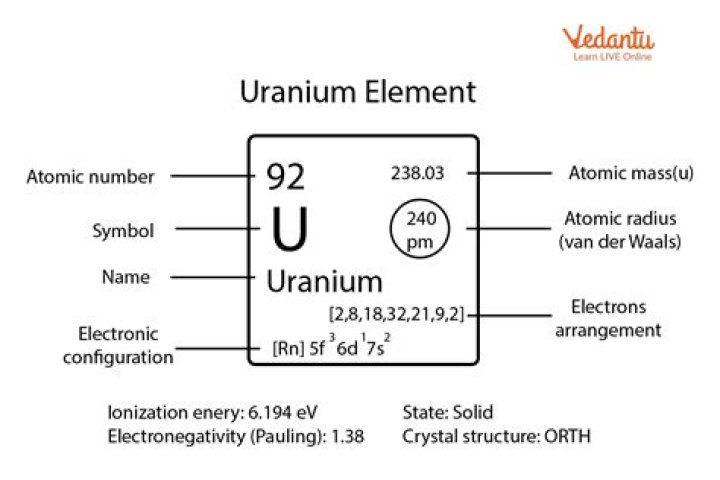
Uranium metal has a very high density of 19.1 g/cm3. Uranium dioxide used in nuclear reactors has a density of 10.97 g/cm3, but this value may vary with fuel burnup. Periodic Table Uranium is a naturally-occurring chemical element with atomic number 92 which means there are 92 protons and 92 electrons in the atomic structure.
What is the density of uranium in a nuclear reactor?
Uranium dioxide used in nuclear reactors has a density of 10.97 g/cm3, but this value may vary with fuel burnup. Periodic Table Uranium is a naturally-occurring chemical element with atomic number 92 which means there are 92 protons and 92 electrons in the atomic structure.
What are the physical properties of uranium fuel?
Physical Properties of Uranium 1 Concentration – Uranium ranks 48th among the most abundant elements found in natural crustal rock. 2 Density – Uranium is very dense. At about 19 grams per cubic centimeter, it is 1.67 times more dense than lead. 3 Melting Point – Uranium melts at about 3,818 degrees Celsius (about 6,904 degrees Fahrenheit).



