What happens when sulfur is burned in oxygen?
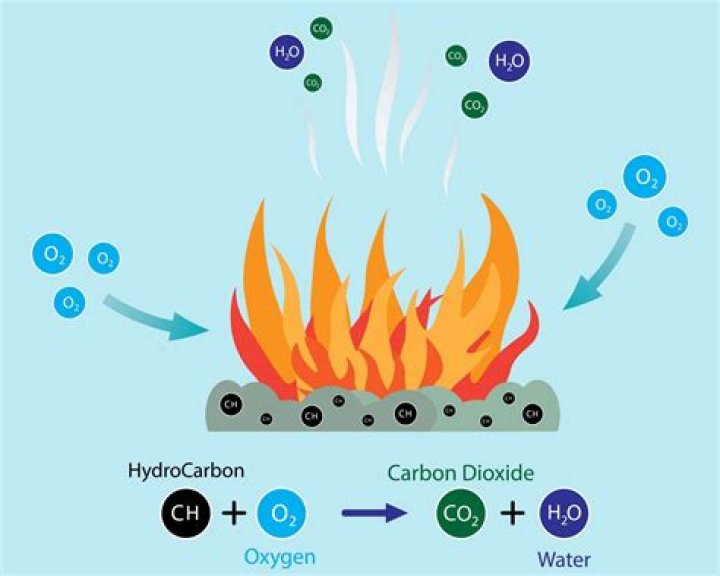
sulfur + oxygen → Sulfur burns in oxygen to form sulfur dioxide. Your teacher will not demonstrate this reaction, because the sulfur dioxide that forms is a poisonous gas that you and your classmates should not be exposed to.
What happens to sulfur when it burns?
When sulfur burns it produces sulfur dioxide (SO2), which turns into sulfurous acid (H2SO3) when it comes in contact with water. That means it can be deadly if you breathe it into your very moist lungs. You’ll know sulfurous acid by another name, too — acid rain.
What happens to sulphur on burning it in air?
Answer: Sulphur when burnt in air forms SO2. Sulphur reacts with oxygen as it burns in the presence of oxygen, producing Sulphur dioxide, which is acidic in nature.
What are 3 interesting facts about sulfur?
Here are some interesting facts about sulfur.
- Atomic number: 16.
- Atomic weight: 32.066.
- Melting point: 388.36 K (115.21°C or 239.38°F)
- Boiling point: 717.75 K (444.60°C or 832.28°F)
- Density: 2.067 grams per cubic centimeter.
- Phase at room temperature: Solid.
- Element classification: Non-metal.
- Period number: 3.
What is burning sulfur used for?
Sulfur is burned or vaporized to control fungus, mites, or insects. When sulfur is burned, it turns into a gas called sulfur dioxide. The gas can mix with moisture on plants to form an acid that can damage plant leaves. Breathing the gas can be harmful to human health.
What is burning sulfur like?
When burned, sulfur produces a blue flame and sulfur dioxide gas — a common pollutant, according to the Environmental Protection Agency. Sulfur dioxide in the atmosphere comes mostly from fossil-fuel power plants and is one of the primary causes of acid rain. The gas is also a lung irritant.
How long should I sulfur burn?
sulphur burners are usually designed for a greenhouse sized room. (1000sq. feet?) 1 hour is enough usually for first burn in a residence.
Does sulfur catch on fire?
Sulfur dust suspended in air ignites easily, and can cause an explosion in confined areas. May be ignited by friction, static electricity, heat, sparks, or flames. Toxic gases will form upon combustion.
Use the name of the product and the general equation given at the start of the chapter to complete the following word equation: Sulfur burns in oxygen to form sulfur dioxide. Your teacher will not demonstrate this reaction, because the sulfur dioxide that forms is a poisonous gas that you and your classmates should not be exposed to.
What are the products of the combustion of sulfur?
The products of the combustion are sulfur dioxide and sulfur trioxide: S (s) + O 2 (g) ——> SO 2 (g) 2S (s) + 3O 2 (g) ——> 2SO 3 (g) These substances react with water in the air to produce sulfurous and sulfuric acid, respectively:
What is the equation for sulfur and oxygen?
Word equation. sulfur + oxygen → sulfur dioxide. Chemical equation. S + O 2 → SO 2. Picture equation
How is sulfur dioxide formed in a reaction?
That is formed as well as the sulfur dioxide. Sulfur has quite a low boiling point, and the heat of the reaction vaporises some of it. What you are seeing is a fog of tiny condensed particles of solid sulfur, as the vaporised sulfur turns back to solid again. Phosphorus