What effect does a lone electron pair have on a molecule shape?
As such, lone pairs of electrons are able to “move” more freely and occupy a larger region of space compared to bond pairs of electrons. As like charges repel, the lone pairs of electrons force the bond pairs of electrons closer to each other and this results in a smaller bond angle within the molecule.
How do lone pair electrons affect the polarity of a molecule?
The middle oxygen atom has a lone pair of electrons and this lone pair gives the molecule its polarity. Lone pair themselves don’t have dipole moment but they do contribute to dipole moment of the molecule.
What effect does the presence of lone pair electrons have on bond angle in a molecule?
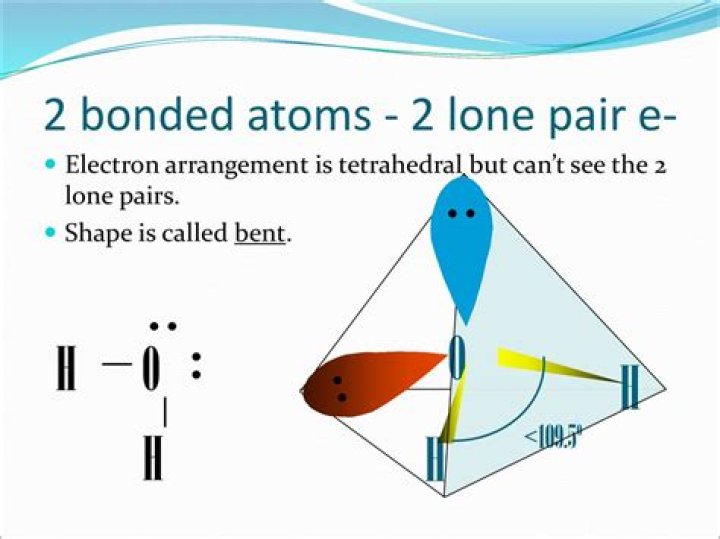
Bond angle is affected by the presence of lone pair of electrons at the central atom. A lone pair of electrons at the central atom always tries to repel the shared pair (bonded pair) of electrons. Due to this, the bonds are displaced slightly inside resulting in a decrease of bind angle.
Do lone pairs make a molecule unstable?
Re: Lone Pairs They must be spread as far apart as possible in order to make the structure more stable (close together causes e- repulsion and therefore instability). Since lone pairs are regions of e- density, you must treat them like another atom attached to the central atom.
Why does a lone pair repel more?
Lone pairs have the greatest repelling effect because they are closer to the nucleus of the central atom compared to the bonding pairs, therefore they repel other lone pairs greater compared to bonding pairs.
How does adding a lone pair affect the position?
Lone pairs will also distort the geometry predicted by VSEPR theory. Lone pairs are not “centered” between to atoms, but tend to “wrap around” the central atom and force the bonding pairs of electrons closer together, thus, causing the distortion in the geometry.
Does having a lone pair make a molecule polar?
Any molecule with lone pairs of electrons around the central atom is polar.
How do lone pairs affect properties?
How the presence of lone pairs affects the bond angles?
Lone pair repulsion: Bond angle is affected by the presence of lone pair of electrons at the central atom. A lone pair of electrons at the central atom always tries to repel the shared pair (bonded pair) of electrons. Due to this, the bonds are displaced slightly inside resulting in a decrease of bond angle.
Why do lone pairs repel more?
Bonding electrons are further away from the nucleus, but they are more localized, so they are not as spread out. This is why two lone pairs will exhibit more repulsion that one lone pair and one bond pair, which in turn will exhibit more repulsion than two bond pairs.
How are lone pair electrons related to bonding electrons?
Thus, the number of lone pair electrons plus the number of bonding electrons equals the total number of valence electrons around an atom. Lone pair is a concept used in valence shell electron pair repulsion theory (VSEPR theory) which explains the shapes of molecules.
How does lone pairs affect the shape of a molecule?
How does lone pairs affect polarity? Lone pair contribute to the shape of the molecule. Clearly, the nucleus ‘end’ of the lone pair is positive and the electron ‘end’ is negative so we can think of a ‘ lone pair dipole’ contributing to the polarity of the molecule in analogy to a bond dipole.
How are lone pair electrons used in VSEPR theory?
Thus, the number of lone pair electrons plus the number of bonding electrons equals the total number of valence electrons around an atom. Lone pair is a concept used in valence shell electron pair repulsion theory (VSEPR theory) which explains the shapes of molecules. They are also referred to in the chemistry of Lewis acids and bases.
How is Lewis electron pair used in chemistry?
The Lewis electron-pair approach can be used to predict the number and types of bonds between the atoms in a substance, and it indicates which atoms have lone pairs of electrons. This approach gives no information about the actual arrangement of atoms in space, however.



