What are the properties of americium?
Chemical properties of americium – Health effects of americium – Environmental effects of americium
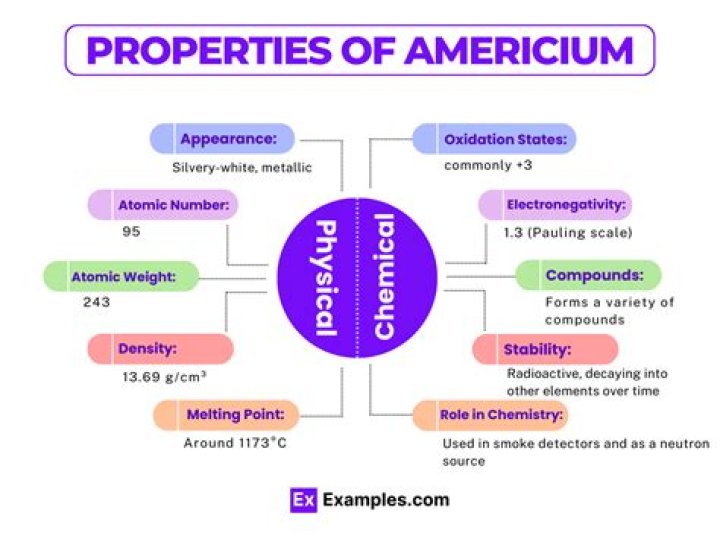
| Atomic number | 95 |
|---|---|
| Density | 13.67 g.cm-3 at 20°C |
| Melting point | 994 °C |
| Boiling point | 2607 °C |
| Vanderwaals radius | unknown |
How is americium-241 used in medicine?
The most common isotope is americium-241. What are the uses of americium-241? Americium also is used as a radiation source in medical diagnostic devices and in research. It is commonly used in minute amounts in smoke detectors as an ionization source.
Is americium man-made?
Americium (chemical symbol Am) is a man-made radioactive metal that is solid under normal conditions. Americium is produced when plutonium absorbs neutrons in nuclear reactors or during nuclear weapons tests.
Why is americium not dangerous?
Since alpha particles do not penetrate the skin and the gamma rays released from americium sources are relatively low in energy, external exposure to americium is not usually considered to be a danger to your health. The radiation from americium is the primary cause of adverse health effects from absorbed americium.
Can you touch americium?
Americium, a silvery-white, synthetic element, is created during nuclear reactions of heavy elements. Americium is a highly radioactive element that can be dangerous when handled incorrectly and can cause severe illnesses.
What makes americium unique?
Americium is a radioactive, soft, silver-white, heavy metal that is denser than lead and slowly tarnishes in air. With a density of about 13.6 g/cc, it is almost twice as heavy as steel. Americium is a transuranic metal, which means it is located after uranium on the periodic table.
Is americium-241 dangerous?
Hazards. Americium-241 has the same general hazards as other americium isotopes: it is both extremely toxic and radioactive. Although α-particles can be stopped by a sheet of paper, there are serious health concerns for ingestion of α-emitters.
Is it legal to own americium?
Americium is the only man-made element which is legal to posses, and the amazing part is that you can buy it for less than $10 at any hardware store.
Do smoke detectors still use americium?
Smoke detectors are very safe Most units sold today use one microcurie or less of americium-241. A 2001 NRC study found people with two of these units in their homes receive less than 0.002 millirems of radiation dose each year.
What americium means?
americium. [ ăm′ə-rĭsh′ē-əm ] Symbol Am A synthetic, silvery-white, radioactive metallic element of the actinide series that is produced artificially by bombarding plutonium with neutrons. Americium is used as a source of alpha particles for smoke detectors and gamma rays for industrial gauges.
Do smoke detectors still used americium?
Ionization chamber smoke detectors contain a small amount of americium-241, a radioactive material. Smoke particles disrupt the low, steady electrical current produced by radioactive particles and trigger the detector’s alarm. Some States require that used smoke detectors be returned to the supplier.
How dangerous is americium?
Is americium still used in smoke detectors?
Smoke detectors are common household items that keep you and your family safe by alerting you to smoke in your home. Ionization smoke detectors use a small amount of radioactive material, americium-241, to detect smoke.
Why is americium-241 safe to use in smoke detectors?
Ionization chamber smoke detectors contain a small amount of americium-241, a radioactive material. Smoke particles disrupt the low, steady electrical current produced by radioactive particles and trigger the detector’s alarm. They react quickly to fires that give off little smoke.
How is americium made?
Americium was first made late in 1944 at the University of Chicago by a team which included Glenn Seaborg, Ralph James, Leon Morgan, and Albert Ghiorso. The americium was produced by bombarding plutonium with neutrons in a nuclear reactor.
Why is americium in smoke detectors?
Ionization smoke detectors use americium as a source of alpha particles. Alpha particles from the americium source ionize air molecules. This makes some particles positively charged and some negatively charged. Because of this shielding, the smoke detector poses no radiation health risk when they are properly handled.