How do lone electron pairs determine molecular shape?
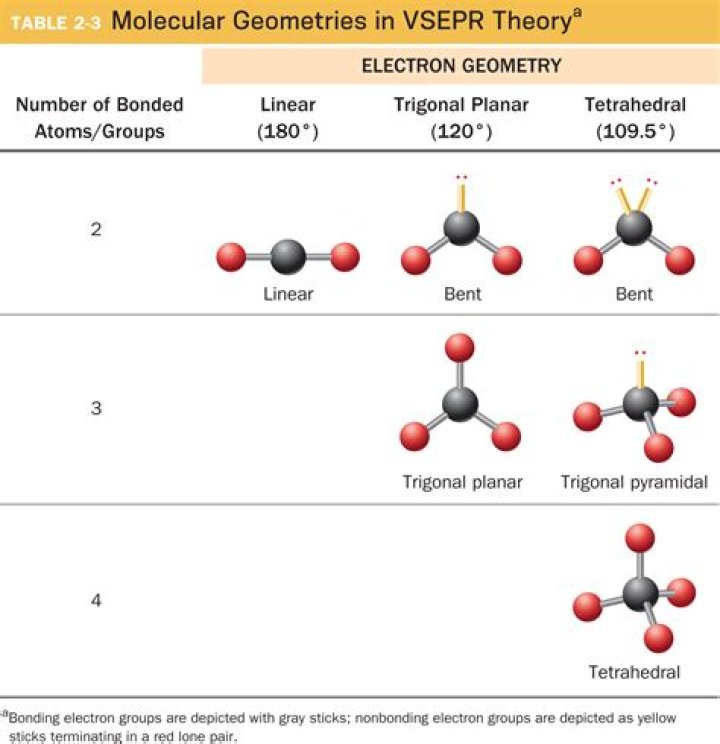
The repulsion between valence electron pairs in the outer shell of the central atom determines the shape of the molecule. You must determine the steric number (SN) — the number of bonding pairs and lone pairs about the central atom. Lone pairs repel more than bond bonding pairs.
What effect does a lone pair of electrons on the central atom have on the molecular geometry?
The molecular geometries of molecules change when the central atom has one or more lone pairs of electrons. The total number of electron pairs, both bonding pairs and lone pairs, leads to what is called the electron domain geometry.
What is the difference between electron-pair geometry and molecular structure?
Explain the difference between electron-pair geometry and molecular structure. Space must be provided for each pair of electrons whether they are in a bond or are present as lone pairs. Electron-pair geometry considers the placement of all electrons. Molecular structure considers only the bonding-pair geometry.
Does nitrogen always have a lone pair?
Carbon tends to form 4 bonds and have no lone pairs. Nitrogen tends to form three bonds and have on e lone pair. Oxygen tends to form two bonds and have two lone pairs. Fluorine (and all halogens) tends to form one bond and have 3 lone pairs.
Does oxygen have a lone pair?
It contains six valence shell electrons and hence, there are three valence electron pairs in an oxygen atom. We know that oxygen forms two bonds and hence two electrons get involved in making those two bonds. So, two electron pairs are left which do not participate in bonding. Hence, oxygen has two lone pairs.
Can two molecules with different electron geometries have the same molecular geometry explain with examples?
In type , when bond pair of electrons is 2 and lone pair of electrons is 2 then the electron domain geometry is Tetrahedral and molecular geometry is Bent. Hence, a molecule having 2 atoms and 2 lone pair have the same molecular geometry as that of the molecule having 2 atoms and 1 lone pair.
How does the number of domains help define the molecular geometry?
The number of electron domains indicates the number of places you can expect to find electrons around a central atom. Lone electron pairs make a major contribution to the shape of a molecule. If there is one lone pair, the result is a trigonal planar shape, while two lone pairs produce a tetrahedral shape.